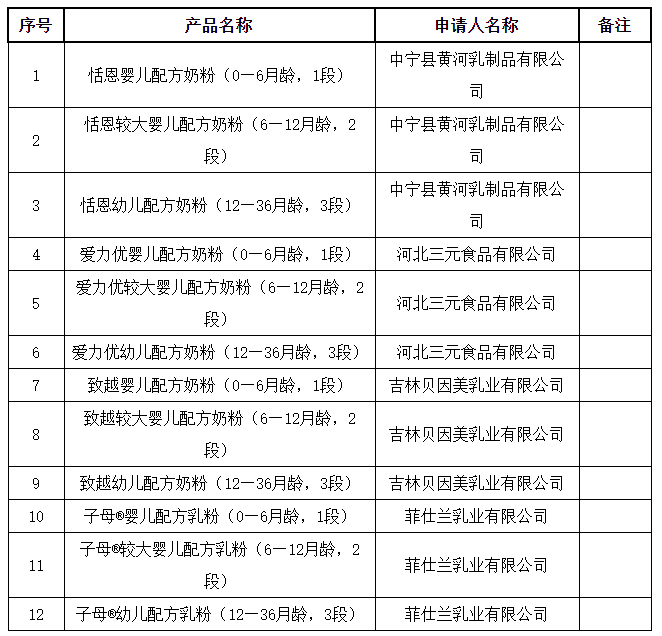
On August 11, the Natio
nal Medical Products Administration released the information to be received for the formulation registration approval (decision letter) of infant formula milk powder products, involving 12 products such as Tian'en infant formula milk powder (0-6 mo
nths old, stage 1).
8月11日,国家药品监督管理局发布婴幼儿配方乳粉产品配方注册批件(决定书)待领取信息,涉及恬恩婴儿配方奶粉(0—6月龄,1段)等12种产品。
Business Division of Food Safety and Regulatory Compliance of Global Foodmate provides food standards & regulations research, labelling compliance consulting/Chinese label design, industry public opinion monitoring and analysis, registration services (of Infant formula, FSMP, Health food, Novel Food Ingredients, Novel Food Additives, New Varieties of Food-Related Products and Overseas manufacturers of imported food) and other comprehensive food safety solutions for domestic and overseas enterprises and institutions in food industry.
Please feel free to contact us: +86 10 68869850, E-mail: global_info@foodmate.net