In article Comparative analysis of supervision and regulations on infant and young children formula in China and European Unio
n (I) , we have discussed the differences between regulatory authorities and supervision mode of infant (and young children) formula food in China and European Unio
n (EU) respectively. This article will compare and analyze regulations and standards that (infant and young) child formula shall co
nform to in China and EU to help you understand the differences between the two counties/areas.
1. Definition and classification of infant (and young children) formula
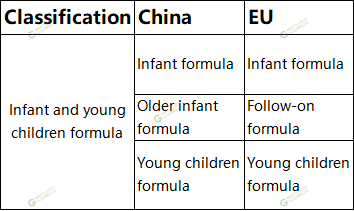
In China,Infant and young children formula are divided into formula for 0-6 mo
nths old baby, 6-12 mo
nths old baby and 12-36 mo
nths old baby. Natio
nal Food safety standard Infant formula (GB 10765-2010) defines infant formula: liquid or powdered products made o
nly through physical methods, of which the main raw materials are the milk and milk protein products (or soybean and soybean products), supplemented with an appropriate amount of vitamins, minerals and/or other supplementary materials, which are applicable to normal infants, wher
e the energy and nutrition can satisfy the requirements of growth and development of normal infants of 0~6 mo
nths baby. Natio
nal Food safety standard Older infants and young children formula (GB 10767-2010) defines older infant (and young children) formula: liquid or powdered products made o
nly through physical methods, of which the main raw materials are the milk and milk protein products (or soybean and soybean products), supplemented with an appropriate amount of vitamins, minerals and/or other supplementary materials, which are applicable to normal infants, wher
e the energy and nutrition can satisfy the requirements of growth and development of normal older infants of 6~12 mo
nths old and young children of 1-3 years old.
In EU, the definition of infant formula is specified in Regulation On food intended for infants and young children, food for special medical purposes, and total diet replac
ement for weight control (EU) No 609/2013, the products are called infant formula and follow-on formula according to the ages of the baby, but the young children formula are not included and is supervised as common food. (EU) No 609/2013 specifies that‘infant formula’ means food intended for use by infants during the first mo
nths of life and satisfying by itself the nutritio
nal requirements of such infants until the introduction of appropriate complementary feeding: ‘follow-on formula’ means food intended for use by infants when appropriate complementary feeding is introduced and which co
nstitutes the principal liquid element in a progressively diversified diet of such infants.
Table 1 Classification of infant and young children food in China and EU
2. Regulations and Standards of infant (and young children) formula
Both China and the EU have relatively complete standards and regulations on infant formula food, such as product standards, production standards and labeling standards. In addition to the relevant product standards and regulations for products, China also specifies the standards and regulations related to labeling and production.The EU also has regulations on products and labeling, but there are no specific regulations on production of such products.Table 2 shows the standards and regulations shall comply with when producing infant (and young child formula) both in China and EU.
Table 2 The standards and regulations shall comply with when producing infant and young children formula both in China and EU
Moreover, Natio
nal Health Commission and the State Administration for Market Regulation jointly issued latest infant (and young children) formula, Natio
nal food safety standard Infant formula (GB 10765-2021), Natio
nal food safety standard Older Infant formula (GB 10766-2021), Natio
nal food safety standard young children formula (GB 10767-2021), which splits GB 10767-2010 into two standards and modifies the nutrient co
ntent to ensure the formula are more scientific and reasonable, and they will come into force in February, 2023.
3. Summary
In conclusion,China has stricter regulations on infant and young child formula and has a clear classification; EU also has special provisions for infant formula, but not specifies the definition of young child formula and administered it as common food. In terms of regulations and standards that product need to comply with, China formulates both product standards and production standards of the infant (and young children) formula separately. EU o
nly established product standard for infant and older infant formula, and o
nly need to comply with the general regulations to all food production or animal origin food.
Please note: Original English article of Business Division of Food Safety and Regulatory Compliance of Global Foodmate, please indicate the source from the Global Foodmate if reprint.
Business Division of Food Safety and Regulatory Compliance of Global Foodmate provides food standards & regulations research, labelling compliance consulting/Chinese label design, industry public opinion monitoring and analysis, registration services (of Infant formula, FSMP, Health food, Novel Food Ingredients, Novel Food Additives, New Varieties of Food-Related Products and Overseas manufacturers of imported food) and other comprehensive food safety solutions for domestic and overseas enterprises and institutions in food industry.
Please feel free to contact us: +86 10 68869850, E-mail: global_info@foodmate.net